2026 AHA Guideline on Acute Pulmonary Embolism
Hey Team,
Big guideline drop a few weeks ago endorsed by an alphabet soup of national specialty bodies and organizations.
This is the first multidisciplinary AHA guideline on acute PE, and ACEP was part of the writing group. The major change is a new clinical severity classification (A–E) designed to guide disposition and escalation of therapy.
1. New Risk Classification (Most Important Takeaway)
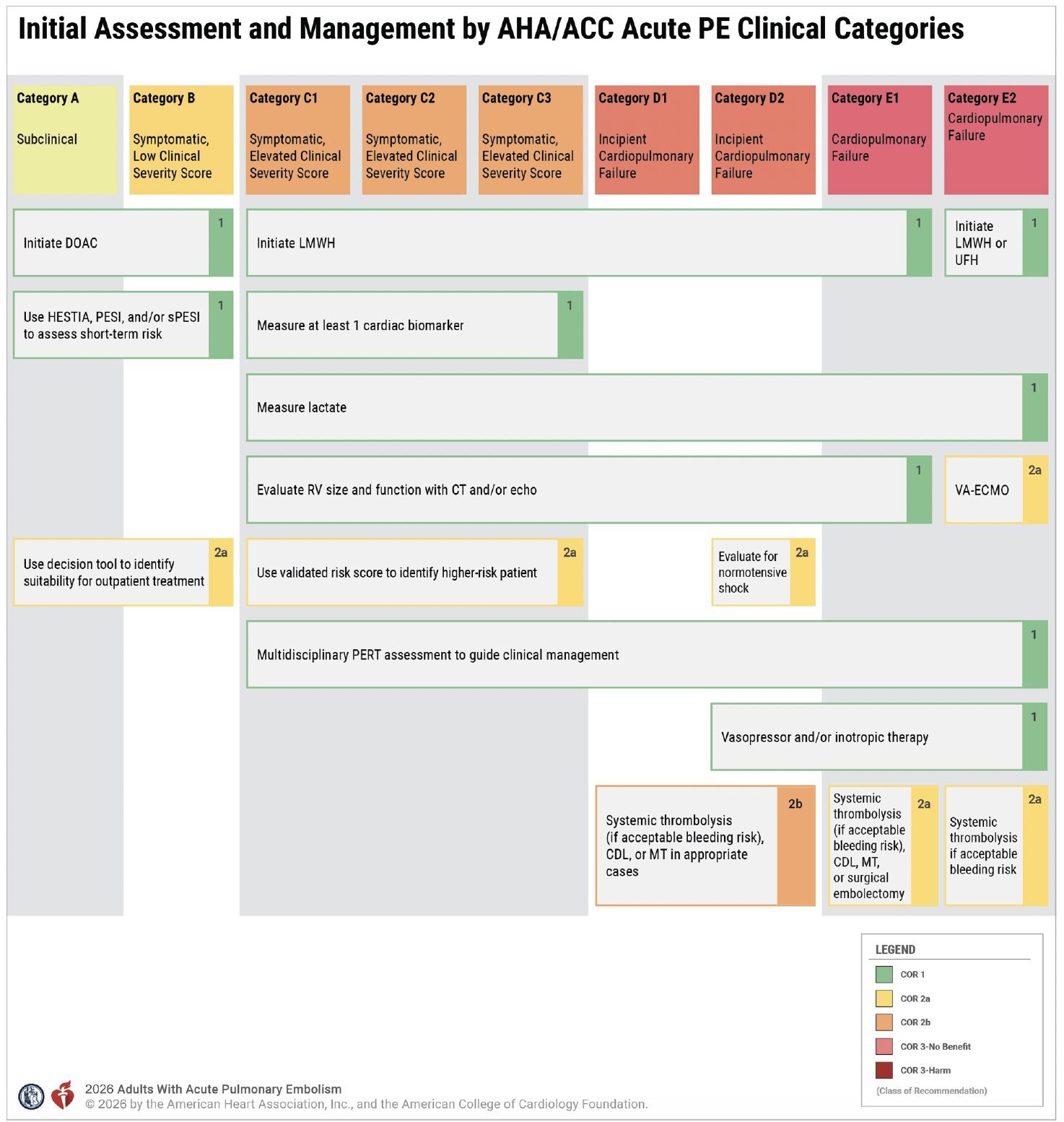
PE severity is now categorized A–E, with escalation in monitoring and therapy as severity increases. If there is hypoxemia or tachypnea there is also a respiratory modifier, “R”.
Category A – Incidental / Asymptomatic
Incidentally diagnosed PE
No symptoms or physiologic impact (Hestia <1 or sPESI <1)
Disposition: outpatient management reasonable
Category B – Symptomatic, Low Clinical Severity
Stable vitals
Low risk by tools (Hestia <1 or sPESI <1)
B1: subsegmental PE
B2: non-subsegmental PE
Disposition: Many patients safe for outpatient treatment or early discharge
Category C – Symptomatic with Elevated Clinical Severity
sPESI ≥1 or Hestia ≥1
Evidence of RV strain or positive biomarkers
Disposition: Admit to hospital. These patients are most at risk of deterioration in the first 24–72 hrs
Subcategories incorporate:
RV dysfunction on CT or echo
Elevated troponin or BNP
Respiratory compromise
Category D – Pre-Cardiopulmonary Failure
Early shock physiology but not persistently hypotensive
D1: transient or recurrent hypotension
D2: normotensive shock (eg lactate >2, AKI, hypoperfusion)
Disposition: ICU level care with consideration of advanced therapies
Category E – Cardiopulmonary Failure
Persistent hypotension, shock, or cardiac arrest
Subgroups:
E1: shock
E2: refractory shock or cardiac arrest
Disposition: ICU with initiation of immediate advanced therapies
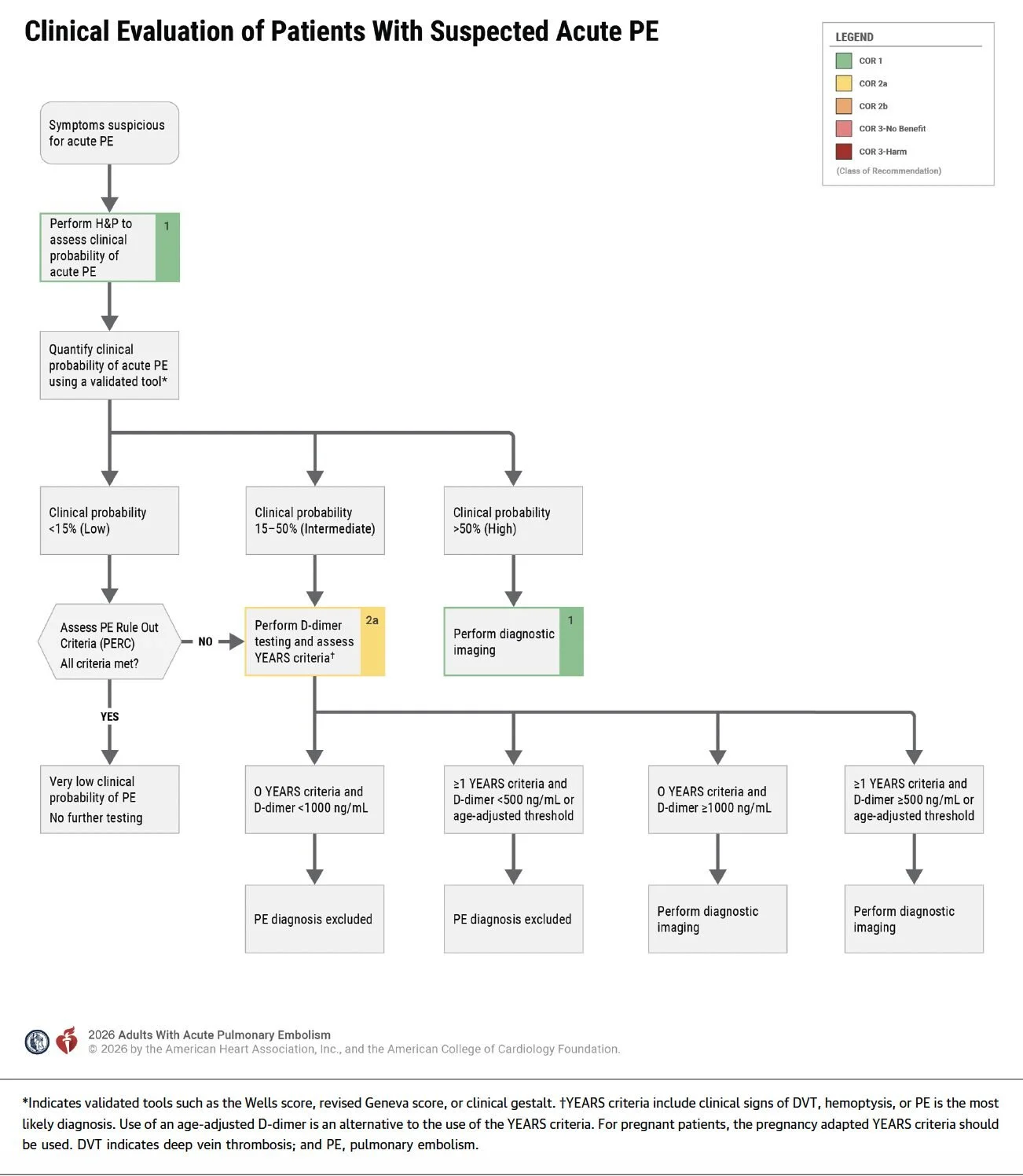
2. Diagnostic Updates
Continue to use clinical decision tools to help estimate probability
Use: Wells, Revised Geneva, PERC
Age-Adjusted D-Dimer and YEARS Criteria effectively exclude PE when applied to patients with low or intermediate clinical probability. AHA now endorses a D-dimer threshhold of 1000 (0 YEARS Criteria) or the 500/age-adjusted D-dimer (if 1 or more YEARS Criteria).
CTPA remains the primary diagnostic test and there are very few reasons at this point to entertain V/Q Scanning.
Excellent diagnostic performance
Less radiation than historically
CTPA is acceptable in pregnancy
RV/LV ratio >1.0 on CTPA predicts higher mortality and deterioration.
3. Risk Stratification Tools
Recommended tools: Hestia, sPESI, PESI, BOVA
For ED use, Hestia or sPESI are the easiest (in my opinion).
Category A-B: sPESI <1, Hestia <1
Category C-E: sPESI ≥ 1, Hestia ≥1
Important limitation: These scores generally predict who will do well. Unfortunately, it is more challenging to determine which hemodynamically stable PE will do poorly and none of these scores can really predict who will deteriorate.
4. Biomarkers
Recommended labs: Troponin, BNP, Lactate
Lactate might be a new biomarker for you in the world of PE. However, it is especially useful in normotensive patients to detect early shock physiology. Elevated lactate (>2 mmol/L) correlates with early complications and deterioration.
5. Imaging for Risk Stratification
All about identifying RV function. There is a deeper dive in the article.
Echo findings suggesting RV dysfunction:
RV/LV >0.9
TAPSE <1.6 cm
McConnell sign
Septal bowing
Echo may outperform CT for short-term mortality prediction. POCUS is backed by the AHA as reasonable if formal echo unavailable.
Thrombus burden on imaging has NOT been shown to be predictive but could help with deciding the appropriate treatment
6. Anticoagulation
LMWH preferred over UFH for most patients who require parenteral anticoagulation (C-E).
More predictable pharmacokinetics
Less monitoring
Lower complication rates
More rapid and consistent achievement of therapeutic levels
DOACs recommended if the patient can take oral meds.
DOACs should be used in those with CKD or liver disease.
In pregnancy or breastfeeding patients, use LMWH or UFH.
7. Hemodynamic Management
Vasopressor of choice: norepinephrine
Benefits:
Raises SVR
Minimal increase in pulmonary vascular resistance
If escalating doses needed:
Add vasopressin rather than pushing norepinephrine too high as higher doses of norepi will start impacting PVR.
If category C2-E, the use of inhaled pulmonary vasodilators may be considered (nitric oxide, epoprostenol)
Fluids: Use small cautious boluses. Excess fluid worsens RV failure.
8. Mechanical Ventilation
AHA recommends to avoid if possible given risk of hemodynamic collapse. If you need to intubate, be sure you have vasopressors, inotropes, and/or VA-ECMO to support the patient in case they become unstable
9. Advanced Therapies
Systemic Thrombolytics we as ED physicians can administer.
Low-dose thrombolysis (25–50 mg tPA) may reduce bleeding risk but evidence is still evolving.
Catheter Directed Thrombolysis is the administration of a thrombolytic drug (most commonly TPA) via a multi-side-hole transcutaneous pulmonary catheter to dissolve a PE.
Mechanical Thrombectomy is a form of percutaneous therapy for acute PE in which a catheter is directed to the location of an indwelling thrombus in the pulmonary arterial system to extract thrombus directly from the pulmonary circulation and externalize it from the body.
Surgical embolectomy involves cardiopulmonary bypass and a sternotomy to extract the clot
The data suggesting which to use is still limited and dependent on a host of patient-specific factors. This is where a PERT team is helpful in making care decisions.
10. PERT Teams
The guideline strongly recommends Pulmonary Embolism Response Teams (PERT) for moderate to severe PE. As you can see from the advanced therapies section, there are a lot of options and a lot of factors to consider when deciding the best care for the patient in front of you.
They function similarly to STEMI teams or Stroke teams
Benefits:
Rapid multidisciplinary decision making
Faster initiation of advanced therapies
11. Post-PE Management
In the first week after discharge, patients should have clinical followup. This is on us to coordinate through discussions with medicine or through expedited referrals.
By the 3 month visit, they should meet to discuss duration of anticoagulation
Up to 50% of patients have persistent dyspnea after PE, even after 3 months of anticoagulation.
This spectrum is called Chronic Thromboembolic Pulmonary Disease (CTEPD). Patients should be screened for symptoms for at least one year.
Bottom Line
The new AHA guideline shifts PE management toward a severity-based framework (A–E) that directly informs disposition and escalation of therapy.
In practical terms:
A–B: outpatient or early discharge on DOACS
C: admit for monitoring (risk of deterioration), LWMH for anticoagulation
D: ICU level monitoring, LWMH, consider advanced therapies
E: ICU level monitoring, LWMH, Immediate thrombolysis or thrombectomy
Also remember:
Use your clinical prediction tools (HESTIA, sPESI) to help decide on the risk category.
Use YEARS or age-adjusted D-dimer to rule out PE
LMWH and DOACs preferred anticoagulation
Lactate is useful to detect early shock
PERT teams recommended for moderate-severe PE